CALIFORNIA
Certifications:

ISO 9001:2015 (C2022-03794) – Quality Management System:
- Demonstrates our dedication to maintaining a robust quality management system to meet customer and regulatory requirements.

Registrations:

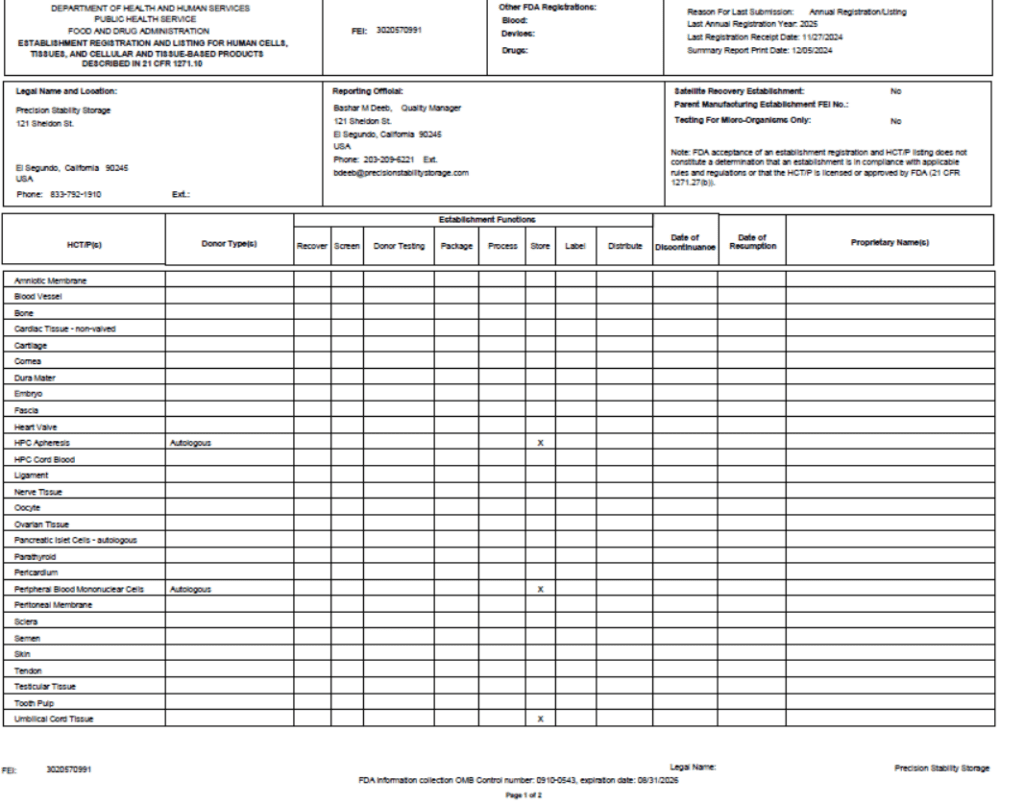
FDA registered Tissue License (3020570991)
- As a registered facility with the U.S. Food and Drug Administration, we comply with regulatory requirements for storing biological products.
FLORIDA
Certifications:

ISO 9001:2015 (C2022-03459) – Quality Management System
- Demonstrates our dedication to maintaining a robust quality management system to meet customer and regulatory requirements.

Registrations:

FDA registered Tissue License (3021868091)
- As a registered facility with the U.S. Food and Drug Administration, we comply with regulatory requirements for storing biological products.

MASSACHUSETTS
Certifications:

ISO 9001:2015 (C2022-05435) – Quality Management System
- Demonstrates our dedication to maintaining a robust quality management system to meet customer and regulatory requirements

Registrations:

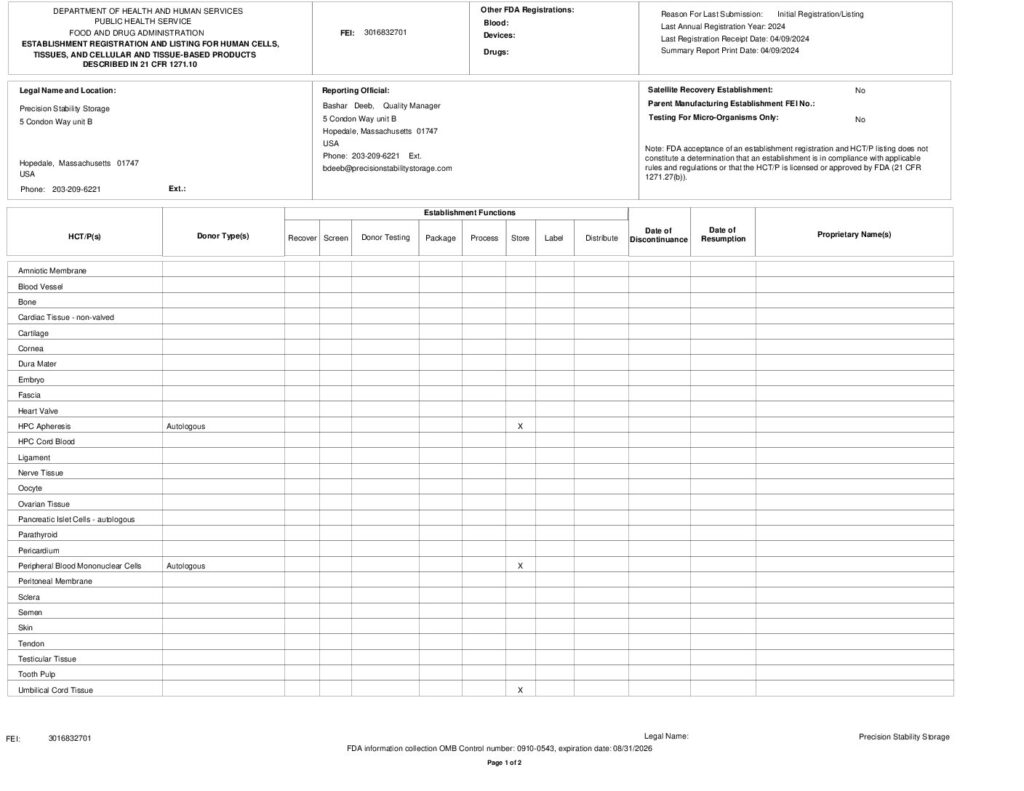
FDA registered Tissue License (3016832701)
- As a registered facility with the U.S. Food and Drug Administration, we comply with regulatory requirements for storing biological products.
NORTH CAROLINA
We undergo audits compliant with ISO 9001:2015, 21 CFR 210, 211, and 820 standards.
Registrations:

FDA Registered – 3007855582
- As a U.S. Food and Drug Administration registered establishment, we comply with regulatory requirements for storing pharmaceuticals and biological products.
Specialized Certifications:
ICH Q1A Stability Testing Compliance:
- Ensures that our stability storage conditions align with the guidelines outlined in ICH Q1A for stability testing.
GxP Regulations:
GMP (Good Manufacturing Practice):
- Adhering to GMP guidelines per customers’ requirements, ensuring our facilities and processes consistently produce and store products to the highest quality standards.
GCP (Good Clinical Practice):
- Adhering to GCP guidelines per customers’ requirements, ensuring clinical trial participants’ safety, rights, and well-being and credibility of clinical trial data.
GTP (Good Tissue Practice):
- Adhering to GTP guidelines per customers’ requirements, aiming to ensure the quality and safety of human tissue used for transplantation, protecting donors and recipients.
GLP (Good Laboratory Practice):
- Adhering to GLP guidelines per customers’ requirements, ensuring the reliability and integrity of non-clinical laboratory studies. Setting forth guidelines for conducting studies and documenting results while providing a framework for regulatory acceptance of data.









